Why Chronic Pain Gets Worse Over Time
Central Sensitization, NMDA Receptors, Glial Cells, and What This Means for Treatment
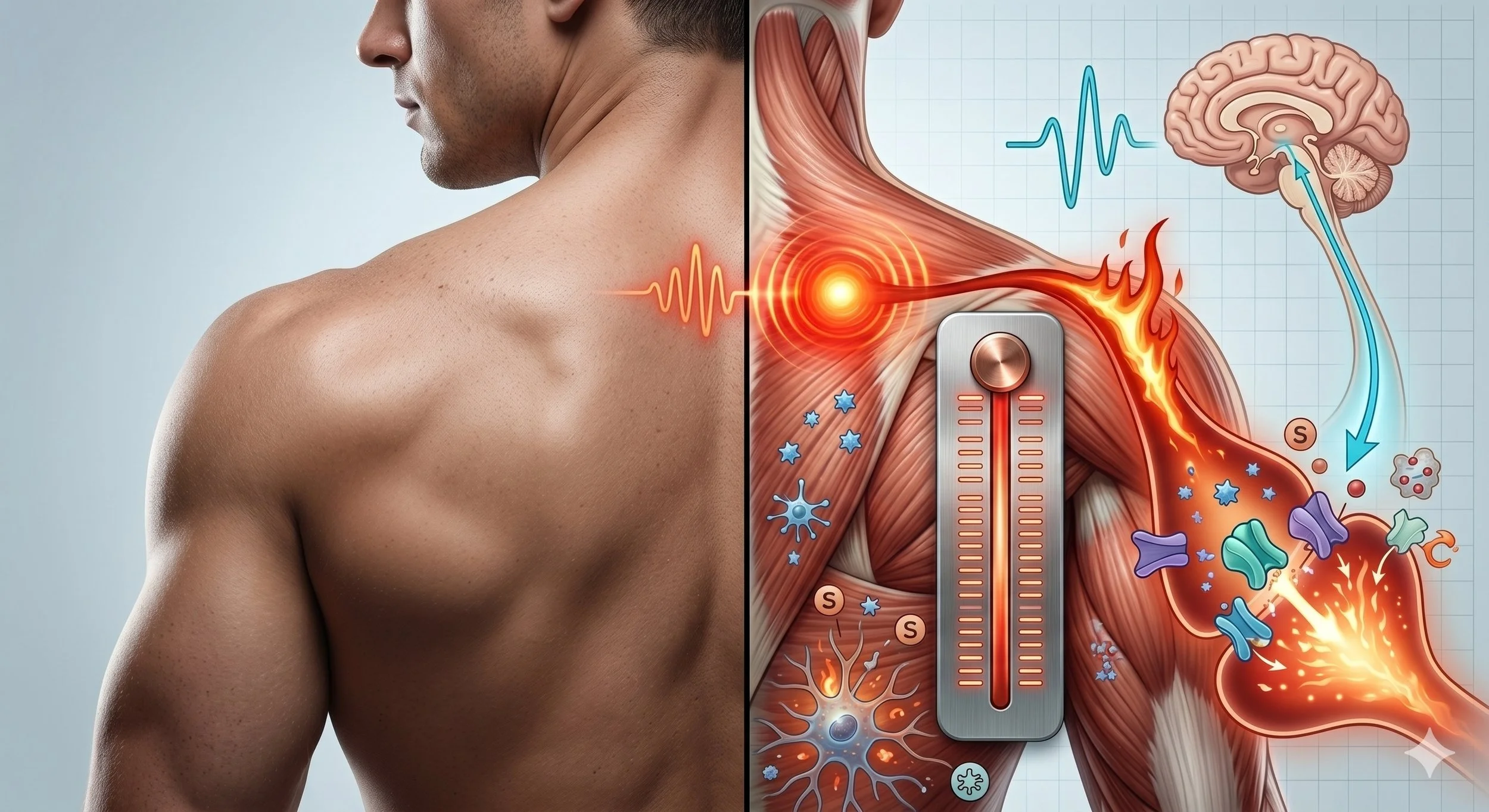
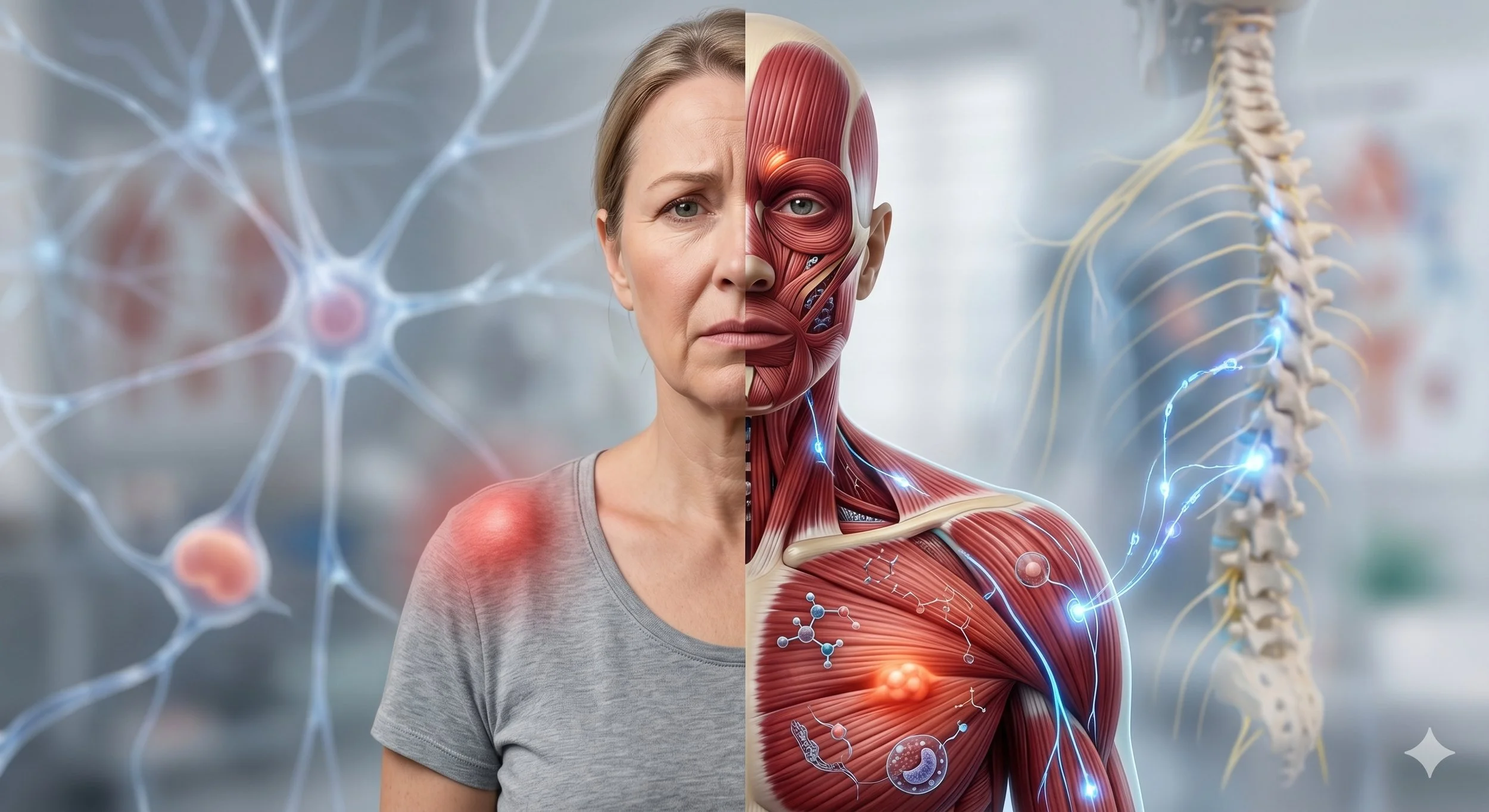
Pain that persists long enough does not simply continue at the same intensity. The nervous system actively amplifies it. Synapses become more efficient at transmitting pain signals, receptors change their molecular identity, and support cells in the spinal cord shift from passive bystanders to active participants in the pain cycle. Understanding these mechanisms explains why chronic muscle pain behaves so differently from acute injury, and why certain treatments can still help.
- Central sensitization is an enhancement in the function of neurons and pain circuits caused by increased membrane excitability, greater synaptic efficiency, and reduced inhibition. It is fundamentally different from peripheral sensitization in both mechanism and manifestation (Latremoliere & Woolf, 2009).
- The NMDA receptor is central to the process: normally blocked by magnesium, it opens when sustained nociceptive input co-releases glutamate, substance P, and CGRP, allowing calcium to enter and initiate intracellular pathways that maintain sensitization.
- Bradykinin receptors can switch from B2 (transient calcium response, less sensitizing) to B1 (prolonged calcium elevation, sustained sensitization) during inflammation. If this switch persists after inflammation resolves, it may mark the transition from acute to chronic pain (Marceau et al., 2002; Schaefers et al., 2003).
- Glial cells in the spinal cord, once considered passive support structures, actively amplify pain by releasing inflammatory mediators and can drive tolerance to opioid medications.
- Conditioned pain modulation (the inhibition of one pain by a second noxious stimulus) may be one mechanism through which dry needling produces analgesia (Bjorkedal & Flaten, 2012).
- Central sensitization produces three measurable clinical phenomena: hyperalgesia (exaggerated pain response), allodynia (pain from normally painless touch), and secondary hyperalgesia (pain spreading beyond the injury site).
Has Your Pain Changed Character or Spread Over Time?
When pain becomes more sensitive, spreads to new areas, or responds to lighter and lighter touch, central sensitization may be involved. Our practitioners assess both peripheral and central components to design treatment that addresses both.
Schedule NowYour Nervous System Has a Volume Knob, and Pain Turns It Up
Central sensitization is not just "more pain." It is a fundamental change in how your nervous system processes sensory information. Latremoliere and Woolf defined it as an enhancement in the functional status of neurons and circuits in nociceptive pathways caused by increases in membrane excitability, synaptic efficacy, and reduced inhibition. The net result is that previously subthreshold synaptic inputs get recruited into nociceptive neurons, generating increased output from the same amount of input (Latremoliere & Woolf, 2009).
Crucially, this means pain is no longer proportional to what is happening in your tissues. With central sensitization, the pain response becomes decoupled from the peripheral stimulus. Light touch can produce severe pain (allodynia). A mildly painful stimulus produces an exaggerated response (hyperalgesia). And pain spreads to areas beyond the original injury (secondary hyperalgesia, which manifests clinically as referred pain).
To trigger central sensitization, the nociceptive input must be intense, repeated, or sustained. A single brief stimulus like a pinch or needle stick is insufficient. Input from many nerve fibers over tens of seconds is required. However, tissue injury is not necessary. This is important because trigger points produce exactly the kind of sustained, ongoing nociceptive input that can drive central sensitization without any visible tissue damage on imaging.
Part 3 Why Does Muscle Pain Spread? Referred Pain and Peripheral SensitizationThe NMDA Receptor Cascade: How Synapses Learn to Amplify Pain
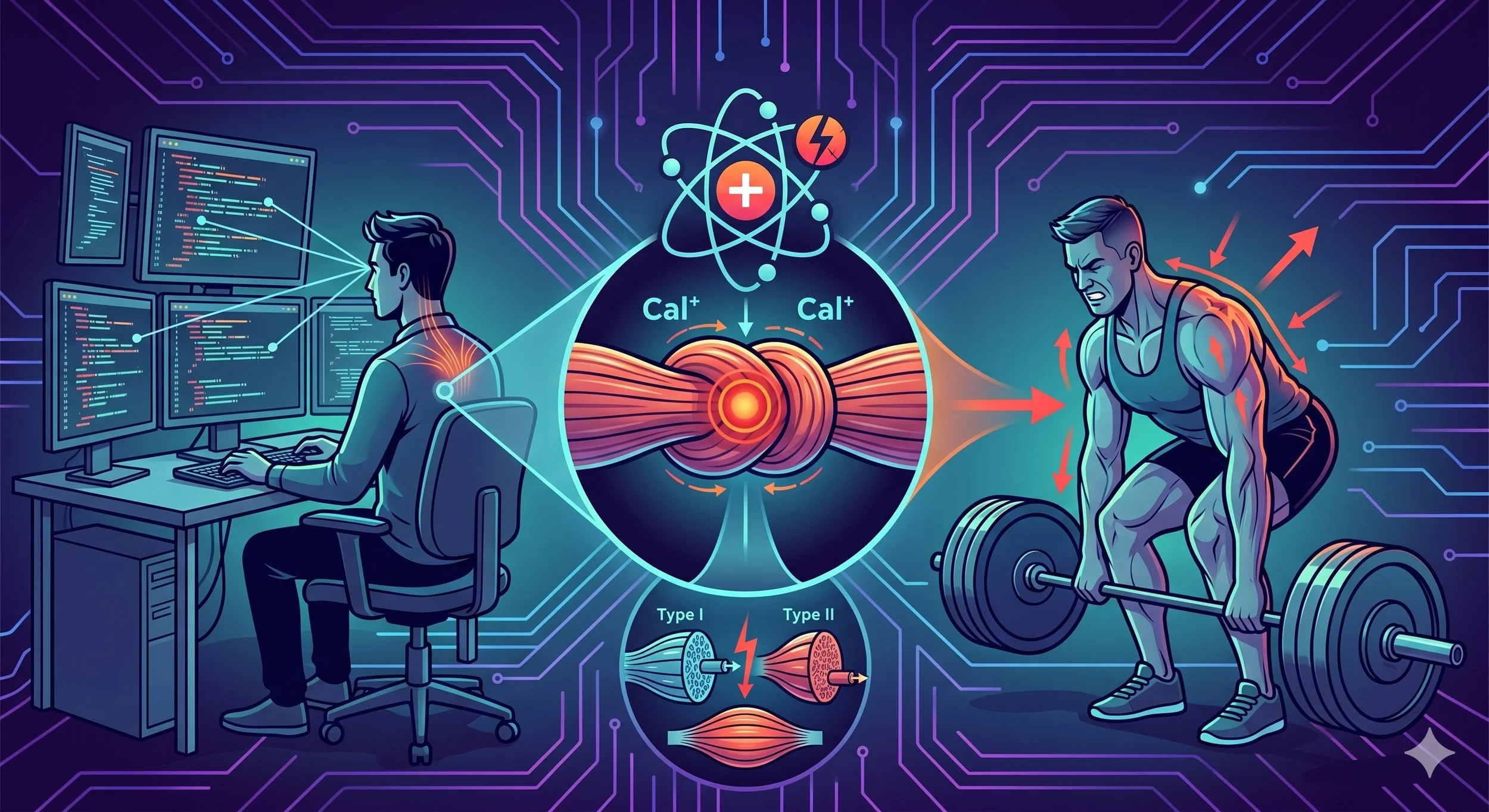
At the molecular level, central sensitization depends critically on a receptor called NMDA (N-methyl-D-aspartate). Under normal conditions, NMDA receptors on dorsal horn neurons are blocked by magnesium ions sitting in the channel pore. Brief, low-intensity pain signals are handled entirely by AMPA receptors, which process the input and let it pass without amplification.
When nociceptive input becomes sustained, the picture changes. Repeated firing of C-fiber nociceptors co-releases glutamate (the primary excitatory neurotransmitter), substance P, and CGRP into the dorsal horn synapse. Substance P binds to NK1 receptors and produces a long-lasting membrane depolarization. This sustained depolarization dislodges the magnesium block from the NMDA receptor, allowing calcium to flow into the cell.
That calcium influx is the critical event. It initiates a cascade of intracellular signaling pathways, including activation of protein kinases (PKA, PKC, CaMKII) and ERK (extracellular signal-regulated kinase), that produce lasting changes in how the synapse operates. ERK maintains central sensitization by boosting NMDA receptor activity, increasing the number of AMPA receptors inserted into the cell membrane (making the synapse more responsive to future input), decreasing potassium channel activity (increasing membrane excitability), and increasing overall synaptic efficiency (Latremoliere & Woolf, 2009; Costigan & Woolf, 2000).
The result is a synapse that has been physically remodeled to transmit pain more efficiently. Previously ineffective synaptic connections become effective. The cell now responds to brief noxious input that it would have previously ignored. This is not a temporary state. It represents a form of neural learning, structurally similar to the long-term potentiation that underlies memory formation in other brain regions.
| Stage | What Happens | Clinical Consequence |
|---|---|---|
| Normal | AMPA receptors handle brief pain signals; NMDA blocked by magnesium | Pain is proportional to the stimulus; light touch feels like light touch |
| Sustained nociceptive input | Glutamate + substance P + CGRP co-released; NK1 receptor activation depolarizes membrane | Pain begins to amplify with repeated stimuli (temporal summation / "wind-up") |
| NMDA unblocked | Magnesium dislodged; calcium floods into the neuron | Transition point: the synapse begins remodeling |
| Intracellular cascade | PKC, PKA, CaMKII, ERK activated; new AMPA receptors synthesized and inserted | Synapse becomes permanently more efficient at transmitting pain |
| Sensitized state | Previously subthreshold inputs now activate the neuron; nociceptive-specific neurons become "wide dynamic range" neurons | Allodynia (pain from touch), hyperalgesia (amplified pain), secondary hyperalgesia (pain spreading) |
When Receptors Change Identity: The B2-to-B1 Switch
One of the most clinically significant mechanisms for the transition from acute to chronic pain involves bradykinin receptors. In healthy muscle tissue, the B2 bradykinin receptor predominates. When B2 receptors are stimulated, they produce only transient increases in intracellular calcium, making sustained sensitization unlikely (Marceau et al., 2002; Calixto et al., 2000).
During tissue inflammation, the picture changes. An additional receptor type, B1, is newly synthesized in the dorsal root ganglion cell body and transported to the peripheral nerve ending, where it is inserted into the membrane alongside the existing B2 receptors. Unlike B2, the B1 receptor produces prolonged elevation of intracellular calcium when activated, which can drive sustained peripheral sensitization. B1 activation can also trigger production of pro-inflammatory mediators including TNF-alpha and IL-1-beta, creating a self-reinforcing inflammatory loop (Schaefers et al., 2003).
The critical concern is what happens when inflammation resolves. If the B2-to-B1 receptor switch persists even after the inflammatory stimulus has been removed, this maladaptive change may represent the molecular mechanism through which acute pain transitions to chronic pain. The nociceptor has been structurally remodeled to respond more intensely and for longer durations, even in the absence of ongoing tissue damage.
Glial Cells: The Amplifiers Nobody Expected
For decades, glial cells in the spinal cord were considered passive support structures for neurons, providing insulation, nutrients, and structural scaffolding. Research over the past two decades has fundamentally changed this view. Glial cells are now understood to be active participants in pain processing that can cause neurons to become hyperexcitable, amplifying and perpetuating the pain signal.
When activated by nociceptive input (or by viruses, bacteria, and other immune triggers), glial cells release a cocktail of inflammatory mediators including IL-1, TNF, IL-6, reactive oxygen species, nitric oxide, prostaglandins, excitatory amino acids, and ATP. These substances both enhance the excitability of projection neurons (the cells that carry pain signals to the brain) and increase the release of substance P and excitatory neurotransmitters from primary afferent nerve terminals, creating a bidirectional amplification loop.
Perhaps most concerning from a clinical perspective, glial cells play a role in the development of tolerance to opioid pain medications. Opiates like morphine bind to receptors on glial cells, but instead of suppressing their activity, morphine actually activates them, pushing them into a pro-inflammatory state that releases more pain-enhancing mediators. This means that the medication intended to reduce pain simultaneously activates a cellular system that amplifies it, helping to explain why opioid effectiveness often diminishes with long-term use.
Chronic Pain That Has Not Responded to Medication?
When pain persists despite medication, the nervous system itself may be amplifying the signal through central sensitization and glial activation. Dry needling targets the peripheral pain generators that feed this cycle, potentially helping to reverse the amplification process from the bottom up.
Schedule NowConditioned Pain Modulation: How a Second Stimulus Can Reduce Pain
Not all of the body's pain-processing changes worsen the situation. Conditioned pain modulation (CPM) is an endogenous inhibitory mechanism in which one source of pain can suppress another. When a "conditioning stimulus" (a controlled nociceptive input) is applied, it can activate descending inhibitory pathways from the brainstem that suppress early nociceptive processing at the spinal level (Bjorkedal & Flaten, 2012).
This mechanism has direct relevance to dry needling. The controlled nociceptive input produced by needle insertion into a trigger point may function as a conditioning stimulus that activates the body's own pain-suppressing circuitry. Research has shown that if a patient expects the conditioning stimulus to reduce pain, the inhibitory effect is amplified, suggesting that patient education and clinical framing play a meaningful role in treatment outcomes.
Recognizing Central Sensitization in Clinical Practice
Identifying whether central sensitization is contributing to a patient's pain is important because it influences treatment intensity, frequency, and approach. Nijs and colleagues outlined clinical criteria that manual therapists can use to assess for central sensitization in musculoskeletal pain patients (Nijs et al., 2010).
| Category | Signs and Symptoms |
|---|---|
| Sensory hypersensitivity | Increased sensitivity to light touch, bright light, noise, temperature changes, and chemical stimuli (pesticides, medications) beyond what the tissue pathology would explain |
| Pain characteristics | Pain that is disproportionate to the identifiable tissue damage; pain that spreads beyond anatomically logical boundaries; pain that persists long after expected healing times |
| Associated symptoms | Fatigue, unrefreshing sleep, concentration difficulties, swollen feeling in limbs (without measurable swelling), tingling, and numbness |
| Conditions commonly associated | Chronic low back pain, chronic whiplash, TMJ disorders, myofascial pain syndrome, osteoarthritis, rheumatoid arthritis, fibromyalgia, chronic fatigue syndrome, chronic headache, irritable bowel syndrome |
When central sensitization is identified, treatment parameters may need adjustment. A patient with significant central sensitization may initially respond better to lower-intensity stimulation, fewer needles, and shorter treatment sessions, with gradual progression as the nervous system recalibrates. Aggressive treatment in a highly sensitized patient can paradoxically worsen symptoms by adding more nociceptive input to an already overloaded system.
What This Means for Your Treatment
Central sensitization does not mean your pain is "all in your head." It means your nervous system has undergone real, measurable changes in how it processes sensory information. These changes are driven by specific molecular pathways (NMDA receptors, ERK signaling, glial cell activation, receptor switching) that can be influenced by treatment.
The clinical evidence suggests a two-pronged approach: address the peripheral pain generators (trigger points) that feed nociceptive input into the sensitized system, while also managing the central amplification through appropriate treatment dosing, patient education, and, when indicated, pharmacological support for descending inhibition. Dry needling occupies a unique position in this framework because it simultaneously targets the peripheral trigger point and may activate conditioned pain modulation pathways that engage the body's own descending inhibitory systems.
Related Trigger Point Dry Needling NYC: How It Works and What It TreatsComprehensive Pain Assessment at Morningside Acupuncture
At Morningside Acupuncture, we are the highest-rated acupuncture and dry needling clinic in New York City with over 500 five-star Google reviews. Our practitioners evaluate both peripheral trigger points and signs of central sensitization to calibrate treatment intensity and approach. Whether your pain is localized or has become widespread, we design treatment plans grounded in pain neuroscience.
Schedule NowReferences
- Bjorkedal, E., & Flaten, M.A. (2012). Expectations of increased and decreased pain explain the effect of conditioned pain modulation in females. Journal of Pain Research, 5, 289-300. https://doi.org/10.2147/JPR.S33559
- Calixto, J.B., Cabrini, D.A., Ferreira, J., & Campos, M.M. (2000). Kinins in pain and inflammation. Pain, 87(1), 1-5. https://doi.org/10.1016/S0304-3959(00)00335-3
- Costigan, M., & Woolf, C.J. (2000). Pain: Molecular mechanisms. The Journal of Pain, 1(3 Suppl 1), 35-44. https://doi.org/10.1054/jpai.2000.9818
- Latremoliere, A., & Woolf, C.J. (2009). Central sensitization: A generator of pain hypersensitivity by central neural plasticity. The Journal of Pain, 10(9), 895-926. https://doi.org/10.1016/j.jpain.2009.06.012
- Marceau, F., Hess, J.F., & Bachvarov, D.R. (2002). Kinin receptors: Functional aspects. International Immunopharmacology, 2(13-14), 1729-1739. https://doi.org/10.1016/S1567-5769(02)00189-3
- Nijs, J., Van Houdenhove, B., & Oostendorp, R.A.B. (2010). Recognition of central sensitization in patients with musculoskeletal pain: Application of pain neurophysiology in manual therapy practice. Manual Therapy, 15(2), 135-141. https://doi.org/10.1016/j.math.2009.12.001
- Schaefers, M., Brinkhoff, J., Neuber, S., Wiesmuller, G.A., & Sommer, C. (2003). Effect of cytokines on bradykinin-induced sensitization of nociceptors. Pain, 104(3), 579-588. https://doi.org/10.1016/S0304-3959(03)00096-5
Disclaimer: This web site is intended for educational and informational purposes only. Reading this website does not constitute providing medical advice or any professional services. This information should not be used for diagnosing or treating any health issue or disease. Those seeking medical advice should consult with a licensed physician. Seek the advice of a medical doctor or other qualified health professional for any medical condition. If you think you have a medical emergency, call 911 or go to the emergency room. No acupuncturist-patient relationship is created by reading this website or using the information. Morningside Acupuncture PLLC and its employees and contributors do not make any express or implied representations with respect to the information on this site or its use. For any legal interpretation of scope of practice in your state, consult a licensed attorney or regulatory authority.